– Development and evolution of symbiotic cells by co-option of a morphogenetic gene –
Takema Fukatsu (Prime Senior Researcher and Leader of Symbiotic Evolution and Biological Functions Research Group) of the Bioproduction Research Institute (BPRI; Director: Tomohiro Tamura), the National Institute of Advanced Industrial Science and Technology (AIST; President: Ryoji Chubachi), Yu Matsuura (former AIST Technological Trainee of Symbiotic Evolution and Biological Functions Research Group; currently a research fellow at Hokkaido University under the fellowship program of the Japan Society for the Promotion of Science for young scientists), and Yoshitomo Kikuchi (Senior Researcher) of Environmental Biofunction Research Group, BPRI, AIST, have analyzed the developmental process and formation mechanism of bacteriocytes for harboring symbiotic bacteria in the seed bug Nysius plebeius, in collaboration with Toru Miura (Associate Professor) of the Faculty of Environmental Earth Science, the Graduate School of Environmental Science, Hokkaido University (President: Keizo Yamaguchi). It was demonstrated that the bacteriocyte formation is mediated by acquisition of new expression patterns of some homeotic genes involved in morphogenesis—particularly Ultrabithorax—during the insect’s embryonic development.
The origin of bacteriocytes, which are insect cells specialized for harboring symbiotic microorganisms, has been a long-lasting mystery in the evolutionary developmental biology. This study unveiled an important molecular mechanism involved in the formation of the host cells for symbiosis. The results are expected to contribute to understanding of the mechanisms of cell differentiation, the molecular-level mechanisms of symbiosis, and the protection mechanisms against bacterial infections.
The results were published online at 3 a.m. on July 14, 2015 (Japan time) in Proceedings of the National Academy of Sciences USA.
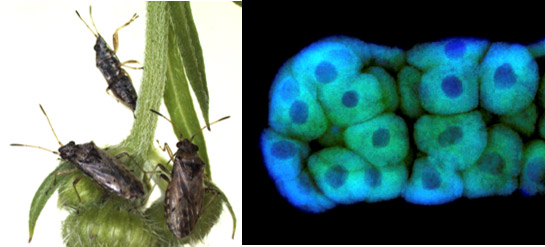 |
The seed bug Nysius plebeius (left) and its bacteriome (symbiotic organ) consisting of a number of bacteriocytes (right)
Green and blue signals indicate symbiotic bacteria and nuclear DNA, respectively. |
Insects account for more than a half of organismal species described thus far in the world. As an animal group representing the biodiversity, the development and utilization of insects’ diverse and sophisticated biological functions are anticipated. Many species of insects are characterized by the capability of harboring microorganisms in their body or even inside their cells, thereby establishing symbiotic relationships. Many insect species, including agricultural and hygienic pests, are associated with intracellular symbiotic bacteria, by which they have acquired essential biological functions such as supply of essential nutrients. The majority of these intracellular symbiotic bacteria are localized within bacteriocytes (cells specialized for symbiosis) in the host insect body, where they play important biological roles and are vertically transmitted to eggs or early embryos of the next generation.
Thus, bacteriocytes are important cells underpinning the elaborate symbiotic relationships between insects and bacteria, and are also interesting from the perspective of pest insect control. However, the origin of bacteriocytes has been a long-lasting enigma, and the mechanisms of bacteriocyte formation have been unknown.
AIST has been investigating important biological functions of symbiotic bacteria of insects (AIST press releases on March 26, 2004, June 30, 2007, April 24, 2012, and September 25, 2014) and sophisticated biological interactions between insects and symbiotic bacteria (AIST press releases on October 29, 2002, June 11, 2013, June 21, 2013, and July 1, 2014).
Particularly, as for symbiotic bacteria found within the bacteriocytes of insects, there have been notable research achievements including “Discovery of the intracellular symbiotic bacterium Wolbachia supplying essential nutrients to bedbugs” (AIST press release on December 22, 2009) and “Capturing the moment of vertical transmission of essential symbiotic bacteria to the next host generation!” (Research results announced on May 28, 2009).
In this study, the researchers investigated the mystery of bacteriocyte formation using the seed bug N. plebeius. This insect, on which very few studies have been conducted, possesses bacteriocytes and its gene function can be analyzed using RNA interference.
Most of the present results were obtained by Yu Matsuura, who was a graduate student at the University of Tsukuba (an AIST Technological Trainee and a research fellow under the fellowship program of the Japan Society for the Promotion of Science for young scientists) in his PhD research project under the supervision of Takema Fukatsu. This study was supported by, in part, a Grant for Science Research from the Ministry of Education, Culture, Sports, Science, and Technology.
In general, stinkbugs are associated with symbiotic bacteria in their gut. However, the researchers have identified that, exceptionally, the seed bug N. plebeius harbors intracellular symbiotic bacteria called Schneideria within the bacteriocytes. The distribution of the symbiotic bacteria in N. plebeius was investigated. Immediately after egg laying, the bacteria were aggregated in the anterior pole of the eggs (Fig. 1A). In mature embryos, the bacteria were localized within a pair of bacteriomes, one on each side of the abdomen (Fig. 1B), where they remained throughout larval and adult stages (Figs. 1C and D).
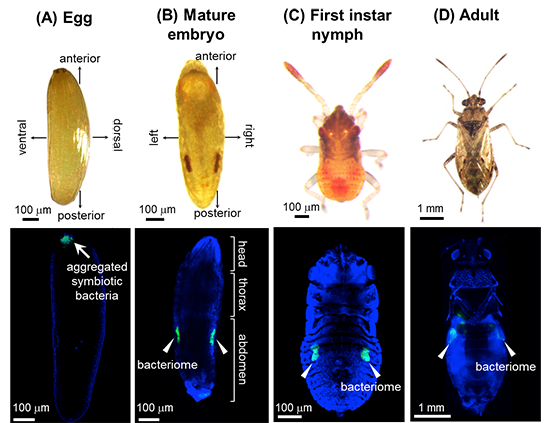 |
Figure 1: Distribution of symbiotic bacteria and bacteriomes in N. plebeius (A) Egg. (B) Mature embryo. (C) First instar nymph. (D) Adult.
Top, bright-field images. Bottom, confocal fluorescence microscopic images. Green and blue signals indicate symbiotic bacteria and nuclear DNA, respectively. |
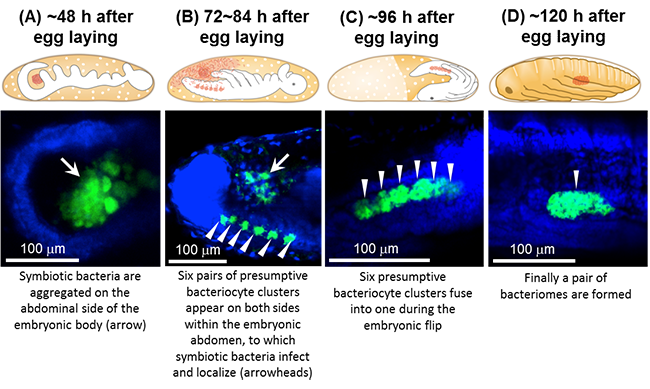 |
Figure 2: Distribution of symbiotic bacteria and bacteriomes in N. plebeius during embryonic development
(A) About 48 hours after egg laying. (B) 72 to 84 hours after egg laying. (C) About 96 hours after egg laying. (D) About 120 hours after egg laying.
Top, schematic illustrations of embryonic morphology. The distribution of symbiotic bacteria is shown in red. Bottom,
confocal fluorescence microscopic images. Green and blue signals indicate symbiotic bacteria and nuclear DNA, respectively. |
The distribution of the symbiotic bacteria during embryonic development was investigated in further detail. About 48 hours after egg laying, the symbiotic bacteria were aggregated at the abdominal outside of the embryo (Fig. 2A). Then, 72 to 84 hours after egg laying, six pairs of aggregated presumptive bacteriocytes appeared on both sides within the embryonic abdomen. The bacteria moved into and infected these bacteriocytes, where they became localized (Fig. 2B). About 96 hours after egg laying, the six bacteriocyte aggregates on each side fused (Fig. 2C), thereby forming a pair of bacteriomes within both sides of the abdomen (Fig. 2D).
There was a previous study reporting that homeotic gene products are localized to bacteriocytes of aphids. However, because gene function analysis is difficult in aphids, no further studies have been conducted. In N. plebeius, gene function analysis is feasible using RNA interference. Hence, the researchers attempted to suppress the expression of various homeotic genes during embryonic development of N. plebeius by RNA interference. When the expression of Ultrabithorax gene was suppressed, no bacteriocytes were formed and, losing the destination to localize, the symbiotic bacteria were scattered around the embryonic body (Figs. 3A and 3B). When normal embryos of N. plebeius were investigated by in situ hybridization, the expression of Ultrabithorax was in complete agreement with the locations of the six pairs of presumptive bacteriocyte aggregates on both sides of the embryonic abdomen (Fig. 3C). In Drosophila, Ultrabithorax is involved in morphogenesis of the posterior thoracic segment and the first abdominal segment. In N. plebeius, on the other hand, a new expression pattern of Ultrabithorax has been acquired and involved in the bacteriocyte formation during the embryogenesis.
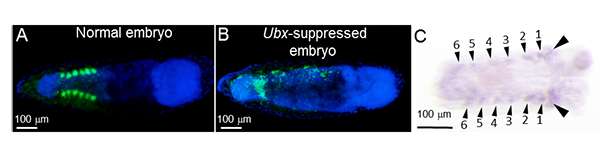 |
Figure 3: Expression of Ultrabithorax in embryos of N. plebeius and the effect of its suppression
(A) Normal embryo: six pairs of aggregated presumptive bacteriocytes are formed in the abdomen, where the symbiotic bacteria are localized.
(B) Ultrabithorax-suppressed embryo: no presumptive bacteriocyte aggregates are formed, and the symbiotic bacteria are scattered.
In (A) and (B), green and blue signals indicate symbiotic bacteria and nuclear DNA, respectively.
(C) Localization of Ultrabithorax expression in normal embryo: six pairs of expression domains (indicated by small arrowheads) are detected,
which are in agreement with the locations of the aggregated presumptive bacteriocytes. Additional expression domains (indicated by
large arrowheads) are found in the posterior thoracic segment and the anterior abdominal segment, which represent the locations
where Ultrabithorax is originally expressed. All the embryos (A)–(C) are about 84 hours after egg laying. |
Other homeotic genes also affect the bacteriocyte formation. In Drosophila, Antennapedia gene controls leg formation. Notably, when the expression of Antennapedia was inhibited by RNA interference, the location of the bacteriomes shifted forward in the embryos of N. plebeius, and, in some embryos, the bacteriocytes were formed within the legs. In Drosophila, the abdominal-A gene controls the formation of the second to eighth abdominal segments. When the expression of abdominal-A was inhibited in the embryos of N. plebeius, the bacteriome fusion became incomplete, being fragmented into several cell aggregates. However, the expression patterns of these genes did not agree with the locations of the six pairs of presumptive bacteriocyte aggregates on both sides of the embryonic abdomen.
In conclusion, while homeotic genes generally play important roles in the development and morphogenesis of Drosophila and other animals, the researchers demonstrated that in N. plebeius, in addition to these general functions, the homeotic genes have been co-opted for the bacteriocyte formation. This study has substantially advanced the understanding of the formation mechanism of bacteriocytes that support the highly integrated symbiosis between insects and bacteria, which has been a long-lasting mystery in the evolutionary developmental biology.
In the next step, the researchers intend to investigate the control mechanisms over the expression of Ultrabithorax for understanding how the expression pattern characteristic of N. plebeius has been acquired. They will seek to understand the whole picture of the gene networks involved in bacteriocyte differentiation by isolating the aggregated presumptive bacteriocytes in embryos of N. plebeius and by performing comprehensive transcriptomic analyses using next-generation DNA sequencers. If signal molecules, by which symbiotic bacteria recognize and infect the aggregated presumptive bacteriocytes in the embryonic development of N. plebeius, can be identified, the achievements may lead to the technologies for controlling infection with symbiotic bacteria in insects, as well as pathogenic bacteria. The researchers will conduct researches from this perspective as well.