- Low temperature reaction and high durability are achieved through a novel ultra porous platinum-alumina catalyst -
Toshihiko Osaki, Senior Researcher, of the Meso-Porous Ceramics Group (Koji Tajiri, Group Leader) of the Materials Research Institute for Sustainable Development (Mamoru Nakamura, Director) and the Advanced Sintering Technologies Group (Koji Watari, Group Leader) of the Advanced Manufacturing Research Institute (Hideto Mitome, Director), both from the National Institute of Advanced Industrial Science and Technology (AIST, Hiroyuki Yoshikawa, Director), together with NGK Insulators Ltd. (Shun Matsushita, President), have succeeded in developing a highly porous platinum-alumina catalyst for use in industrial exhaust gas purificators (Figure 1) with the characteristic of high temperature resistance coupled with a high efficiency.
The reaction temperature of this catalyst is 100 ºC lower than the reaction temperatures of the catalysts currently in use, and the thermal resistance has been improved to approximately 200 ºC. The characteristics of this catalyst, together with the use of low cost aluminum hydroxide as the starting material, and the implementation of a low cost and simple process such as freeze-drying, will lead to the spread of its applications.
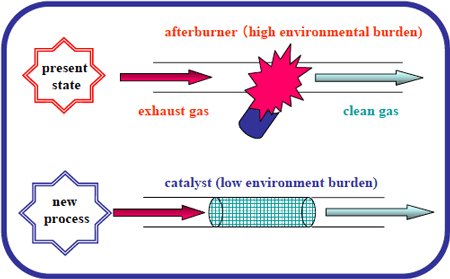
Figure 1 Applications for the new catalyst developed |
|

Photograph 1. Platinum-alumina cryogel catalyst (18 mm in diameter, and 23 mm in length)
|
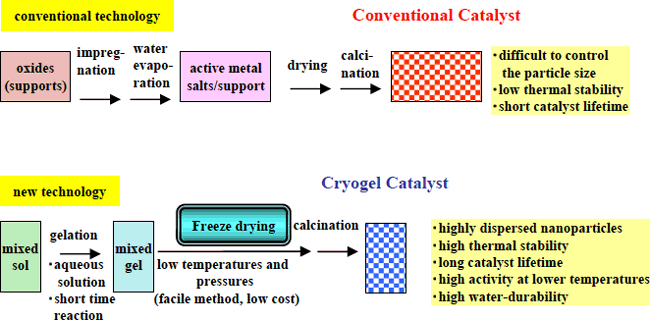
Catalysts containing platinum or other precious metal particles supported on alumina are used for removal by oxidation of the VOC (volatile organic compounds) that are included in the waste gases of factories. Traditionally, these catalysts have been prepared by the impregnation method to easily support the precious metal particles. However, this method has as drawbacks the low dispersibility of the precious metal and the non-homogeneity of the particle diameter. Moreover, as the temperature reaches several hundred degrees during the catalytic reaction, the surface area is reduced by sintering of the particles. In consequence, the catalytic activity is decreased and the lifetime is shortened (Figure 2).
During the fiscal years 2003 through 2005, AIST and NGK Insulators carried out a joint research project "Research on Low Environmental Impact Processes," based on a system of cost burden share ("matching fund system"). As part of this research, the development of a highly efficient catalyst for the purification of VOC gases produced during calcination of ceramics has been pursued. VOC gases are produced by a combustion of polymer binders mixed during preparation of ceramics.
Based on the technology for the manufacture of highly porous materials developed at AIST, a new processing method for a homogeneous platinum-alumina gel was focused on, with freeze-drying chosen as a low cost and simple process to dry this gel (Figure 2).
As a result, not only was improvement of catalytic activity and durability at high temperature achieved, but it was also possible to manufacture a new highly porous platinum-alumina ceramic body with high durability characteristics (Photo 1). As the material is manufactured through low temperature freeze-drying, it has been named "platinum-alumina cryogel."
Boehmite sol, low cost aluminum hydroxide, is the starting material for this cryogel catalyst. Use of chelating agents such as oxalic acid and malonic acid when adding the platinum source to the sol, protects the platinum ion, contributing to suppression of precipitation of platinum black, and producing a homogeneous dispersion of ultra fine platinum particles. As the freeze-drying process is carried out without using solvent for substitution of the damp gel, there is not overflow of precious metal ions.
 |
|
Figure 2 Comparison of current and new technologies |
Removal of methane in air by oxidation was carried out to assess the efficiency of the cryogel catalyst manufactured. As shown in Figure 3, it is possible to achieve sufficient removal at a reaction temperature approximately 100 ºC lower than that of current catalysts.
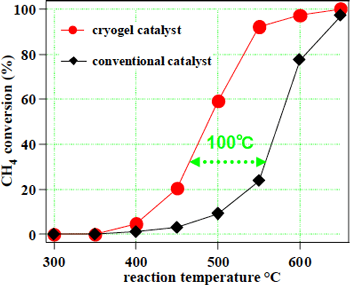
Figure 3 Methane oxidation activity in the platinum-alumina catalyst |
|

Photo 2 Ultra fine platinum particles in the Platinum-alumina Cryogel. Platinum 5-wt%; black spots correspond to ultra fine platinum particles 1 nm in diameter.
|
 |
|
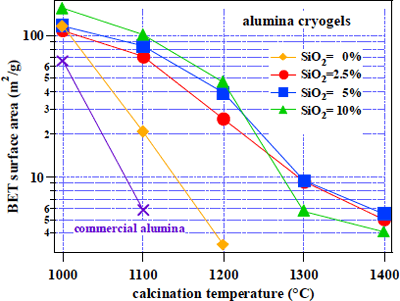
Figure 4 Surface area of alumina cryogel |
Until recently, it was not possible to obtain heat resistance catalysts using traditional manufacturing methods, and sintering of ultra fine particles of platinum occurred. In the case of a cryogel catalyst there is homogeneous distribution of platinum particles of approximately 1 nm (1 nanometer: 1/109 meters) in size (Photo. 2). It is thought that a strong interaction between the cryogel carrier and the ultra fine platinum particles hinders the sintering of the fine particles, imparting a thermal resistance characteristic to the catalyst. This ultra fine structure may also determine that the catalytic reaction advances effectively even at low temperatures.
The changes on the surface area of the alumina cryogel particles according to the calcining temperature are shown in Figure 4. The surface area of commercial alumina decreases rapidly upon calcining, whereas it is evident that the alumina cryogel presents high thermal resistance characteristics. Addition of silica (SiO
2) further improves the thermal resistance characteristics.

Photo 3 Alumina cryogel with 10-wt% silica addition (1200 ºC, 5 hours calcining)
|
|

Photo 4 Commercial alumina (1100 ºC, 5 hours calcining), the scale is approximately 10 times that of Figure 3.
|
Photo 3 corresponds to the TEM image of alumina cryogel with 10-wt% silica addition (1200ºC, 5 hours calcining). Fine-grained alumina particles are observed after calcining at high temperature. For commercial alumina, a coarse sintered structure is already present at 1100ºC. (Photo 4; please take note of the scales in photos 3 and 4). Thus, in this cryogel we can expect not only increased durability of the carrier and of the precious metal particles, but also a long life of fine metal particle so that it is able to withstand long reaction times at high temperature.
The pore structure constitutes most of the volume of the manufactured cryogel, but even when it is a multi-porous body with low bulk density (nearly equal to 0.06g/cm
3), structural destruction by water is not observed (Photo 5). There are no observable changes before and after wetting in the separately measured pore distribution curve, which indicates that it is possible to use a conventional immersion method to support fine catalyst metal particles. These new characteristics cannot be observed in the currently used aerogel which also presents a large volume composed of pores.
 |
|
Photo 5 The newly developed cryogel is stable in water, whereas water causes structural destruction to the aerogel currently in use. |
NGK Insulators plans to carry out purification tests of exhaust gases using the developed platinum-alumina cryogel catalyst in its own furnaces for sintering of ceramics.
A wide range of applications is open to the developed platinum-alumina cryogel catalyst in applications where high porosity of catalyst or catalyst carriers is required.
Research on cryogels has just begun. Further basic and applied research work will be directed towards the development of the synthesis technologies for the large-scale production of platinum-alumina (Pt-Al2O3) cryogel, which is crucial for the spread of its applications. Also, the use of other precious metals such as palladium, rhodium, etc. as well as base metals will be explored, and the mechanism that imparts water resistance will be investigated. Finally, the design of more advanced techniques, such as the dispersion of more than two precious metals in cryogel, will be explored.